| Active Ingredient |
Drug Name |
FDA-Approved Use |
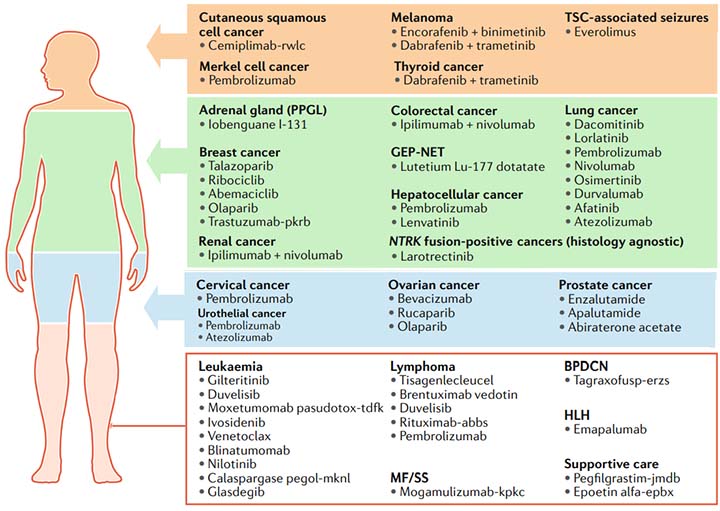
| Pembrolizumab >>
|
Keytruda |
To treat Merkel cell cancer, hepatocellular cancer, lung cancer, cervical cancer, urothelial cancer and lymphoma |
| Nivolumab >>
|
Opdivo |
To treat colorectal cancer and renal cancer in combination with ipilimumab, as well as lung cancer with monotherapy |
| Durvalumab >>
|
Imfinzi |
First-line treatment of patients with stage III non-small cell lung cancer (NSCLC) and metastatic NSCLC |
| Atezolizumab >>
|
Tecentriq |
Indicated for locally advanced or metastatic urothelial carcinoma and metastatic NSCLC |
| Ipilimumab >>
|
Yervoy |
To treat colorectal cancer and renal cancer in combination with nivolumab |
| Rituximab >>
|
Rituxan |
Indicated for untreated and previously treated CD20-positive CLL,combined therapy with fludarabine and cyclophosphamide (FC) |
| Bevacizumab >>
|
Avastin |
Indicated in combination with paclitaxel, pegylated liposomal doxorubicin, or topotecan for women with platinum-resistant recurrent epithelial ovarian |
| Prucalopride >>
|
Motegrity |
To treat chronic idiopathic constipation |
| Gilteritinib >>
|
Xospata |
To treat the relapsed or refractory acute myeloid leukemia (AML) |
| Larotrectinib >>
|
Vitrakvi |
To treat patients whose cancers have a specific genetic feature (biomarker) |
| Glasdegib >>
|
Daurismo |
To treat newly-diagnosed AML |
| Revefenacin >>
|
Yupelri |
To treat chronic obstructive pulmonary disease |
| Lorlatinib >>
|
Lorbrena |
To treat patients with ALK-positive metastatic non-small cell lung cancer |
| Talazoparib >>
|
Talzenna |
To teat locally advanced or metastatic breast cancer patients with a germline BRCA mutation. |
| Dacomitinib >>
|
Vizimpro |
To treat metastatic non-small-cell lung cancer |
| Duvelisib >>
|
Copiktra |
To treat relapsed or refractory chronic lymphocytic leukemia, andsmall lymphocytic lymphoma |
| Elagolix >>
|
Orilissa |
For the management of moderate to severe pain associated with endometriosis |
| Ivosidenib >>
|
Tibsovo |
To treat patients with relapsed or refractory acute myeloid leukemia |
| Encorafenib >>
|
Braftovi |
To treat unresectable or metastatic melanoma |
| Binimetinib >>
|
Mektovi |
To treat unresectable or metastatic melanoma |
| Moxidectin >>
|
Moxidectin |
To treat onchocerciasis due to Onchocerca volvulus in patients aged 12 years and older |
| Baricitinib >>
|
Olumiant |
To treat moderately to severely active rheumatoid arthritis |
| Fostamatinib >>
|
Tavalisse |
To treat thrombocytopenia in adult patients with chronic immune thrombocytopenia (ITP) |
| Apalutamide >>
|
Erleada |
To treat a certain type of prostate cancer using novel clinical trial endpoint |
| Tezacaftor >>
|
Symdeko |
To treat cystic fibrosis in patients age 12 years and older |
| Tenofovir >>
|
Biktarvy |
To treat infection in adults without antiretroviral treatment history or to replace current antiretroviral regimen
|